Summary:
- pH is a measure of the acidity of an aqueous (water-based) solution.
- pH can be used to determine the amount of hydrogen ions in a solution.
- In soil science, pH is important because it affects the solubility of many important plant nutrients and potential toxins.
- pH can be measured in several ways, some more exact than others.
- In the laboratory, soil pH is often measured using a pH probe, which gives an exact reading of the pH, usually to 2 decimal points.
- Measuring soil pH is simple but critical for any investigation into soil health.
Outline:
You’re probably familiar with the concept of pH from an earlier science or chemistry class. Perhaps you’ve already heard that lemons are “acidic” or you’ve seen “alkaline water” on the shelves of your grocery store. pH is extremely important in soil science, but why? Let’s discuss.
What is pH?
Simply put, pH is a measure of the acidity or alkalinity of an aqueous solution. An aqueous solution is basically any liquid mixture in which water is the solvent. In our case, the aqueous solution would be a slurry of soil and water after the solid particles have been allowed to settle to the bottom.
pH is measured on a 14-point scale. A solution is neutral if its pH is around 7. That means that is it neither acidic nor alkaline; i.e., there’s the same amount of acid ions that there are base ions. Acids have a pH below 7, and bases have a pH above 7. Therefore, a solution is acidic if its pH is below 7, and a solution is alkaline if its pH is above 7.
That’s what most people know about pH, but you may be wondering how pH is measured in the first place. There’s debate about what, exactly, pH stands for, but some think of it as the “power of hydrogen” since pH is the negative logarithm of the concentration of hydrogen ions in solution. When you measure pH, you are measuring the number of hydrogen ions in the solution and then converting it to a logarithmic scale. Here are the formulas:
pH = -log([H+])
[H+] = 10-(pH)
Where [H+] is the molarity of hydrogen in solution; i.e., the concentration of H+ ions per liter of solution.
Knowing what, exactly, pH is isn’t important to interpreting the results of a soil pH test, so don’t worry if you don’t understand it fully!
Why is pH so important in soil science?
Ever since the dawn of agriculture, pH has been vital to crop success. You may be familiar with some old agricultural terms that describe the pH of the soil. “Sweet” soils are those soils with a pH that is slightly alkaline. Likewise, “sour” soils are more acidic. These terms have no pH boundaries and are based on more qualitative evidence. Clearly, they originated before we started measuring soil samples in a laboratory. So why is pH so important in soil science?
Many nutrients that are critical to plant health are pH-dependent. Essentially, certain nutrients become more or less available to plants based on changes in pH.
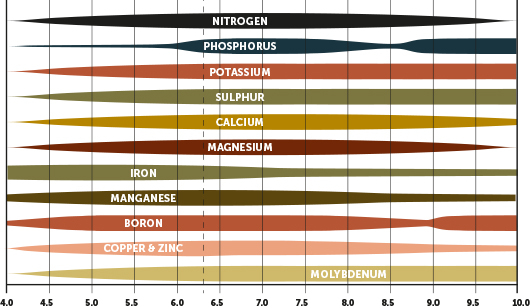
For example, as pH drops below 6, the essential macronutrient phosphorous becomes very difficult for plants to access because it leaves the soil solution. Conversely, below pH 6, iron starts to enter into the soil solution. Too much iron in the soil solution can be toxic for plants.
As a general rule, as soil pH becomes more acidic, the availability of macronutrients goes down, and micronutrients (i.e., potential toxins) goes up. As pH becomes more alkaline, the availability of macronutrients increases, but the availability of micronutrients drops. Soil pH levels between 5.0-7.5 are adequate for most plants.
How do you measure pH?
There are several ways to measure pH. In high school, students are generally taught to measure pH using indicator solutions or pH paper. These tools are inexpensive, but imprecise, ways to qualitatively assess the pH of a sample. Many at-home soil sample kits include one of these methods.
Indicator solutions and pH paper only allow you a general sense of the pH of your soil. While it might be good enough to help you determine where to plant blueberries, it’s not good enough for the lab, and it’s not good enough for a true understanding of your soil.

In soil science laboratories, pH is often measured using a pH probe. These glass probes are created specifically to measure H+ concentration, and do so by measuring the electrochemical potential created across the bulb when it is immersed in solution.
Measuring pH in the lab is a multi-step process. Soil samples are first dried (usually at 55 degrees Celsius) and then crushed or sieved to obtain the fine earth fraction, i.e., the bits that are less than 2mm in diameter, to reduce the presence of rocks.
After drying, some of the sample is combined with deionized water in a 1:1 or 1:2 ratio by weight in a test tube. The sample is then shaken to mix the soil thoroughly with the water and allow all H+ ions present to release into the solution.
While the sample is shaking, the pH probe has to be calibrated. This is done by inserting the probe into buffer solutions–solutions that have a consistent pH–and using the “calibrate” function on the pH meter.
The sample is allowed to settle briefly, then the calibrated pH probe is inserted into the aqueous solution above the settled soil particles. The probe is attached to a machine which gives a pH reading, usually to 2 decimal places. And that’s it! Now you know the pH of your sample, and can use it to inform nutrient management decisions or soil health investigations.
Method source: https://swel.osu.edu/sites/swel/files/imce/pH%20%26%20EC%20SOP%20SHI.pdf